Sinepecten segonzaci Schein, 2006
SCHEIN, E. 2006. A new deep-sea pectinid bivalve from thermal vents of Manus back-arc Basin (south-western Pacific), Sinepecten segonzaci n.gen., n. sp. (Pectinoidea: Pectinidae), and its relationships with the genera Bathypecten and Catillopecten. Zootaxa, 1135: 1-27 [p. 5, figs. 2-12, 13b]
2006 Sinepecten segonzaci Schein, 2006
|
E. Schein, 2006, figures 8, 9, 11.
|
«Sinepecten segonzaci n. sp.
Figures 2–3, 4–12. Locus typicus: western Pacific, Manus back-arc Basin, (north Papua-New Guinea), hydrothermal vent area PACMANUS, Field E, Barnacle site, 03°43.62’S – 151°40.32'E, depth : 1674 m.
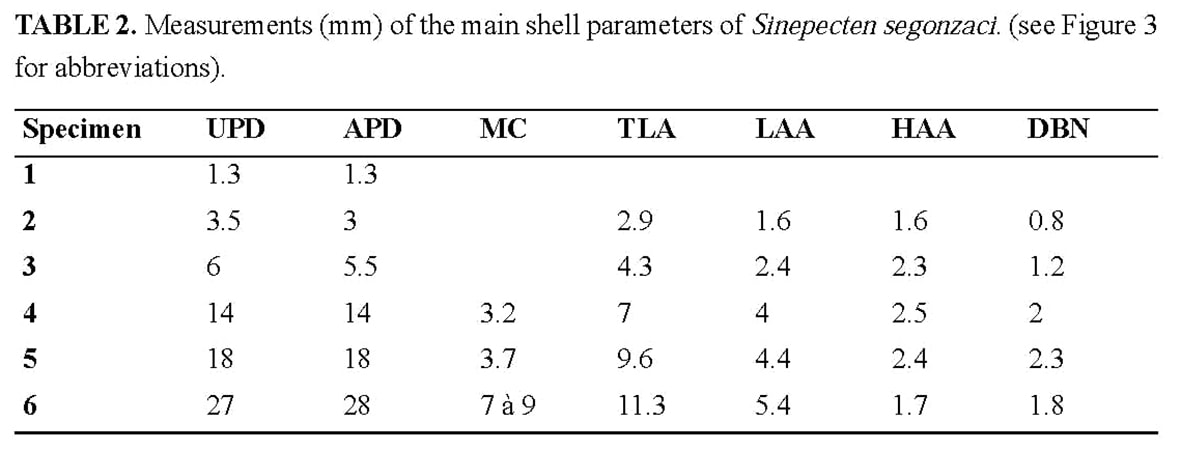
Type material: Type series consisting of five bivalve specimens (specimens 2 to 6) of increasing size, from 3.5 to 27 mm of UPD (Table 2), the largest one (specimen 6) being designated as the holotype. It is catalogued in the National Science Museum of Tokyo with the following registration numbers: NSMT-Mo 73697 (holotype), NSMT-Mo 73698 (specimen 5), NSMT-Mo 73699 (specimen 4), NSMT-Mo 73700 (specimen 3), NSMT-Mo 73701 (specimen 2).
Diagnosis: Shell thin, valves not gaping. Lower (right) valve flat, upper (left) valve convex. Ornamentation commarginal, affecting only the external surface, very fine ridges or lamellae covering the left valve from beak to margin in juveniles and adults, and the right valve only after the juvenile stage. Posterior auricles poorly delimited. Byssal notch without ctenolium under the anterior auricle of the right valve, widely opened in the juvenile, but gradually closing and finally overlapped by the dorsal expansion of its lower edge in the adult. Test principally calcitic, with an aragonite layer restricted to the area inside of the pallial line.
Material examined: The five specimens of the type series, which were collected alive, except specimen 5, come from the type-locality. Two of them (specimens 2 and 4) were treated for SEM observations. The left valve of specimen 5 was treated with Feigl’s solution for the identification of aragonite. A single postlarval valve in relatively poor preservation (specimen 1) was collected with sediment in a site without active venting. This material was collected by the manned submersible Shinkai 2000 during two dives: dive #1075, 22.11.1988, Field E, Barnacle site, 03°43.62’S – 151°40.32'E, 1674 m (type series); dive #1076, 23.11.1998, Field D, slurp gun 2, 03°43.73’S –151°40.21’E, 1623 m (specimen 1). It was fixed in formalin, then preserved in ethanol 80%.
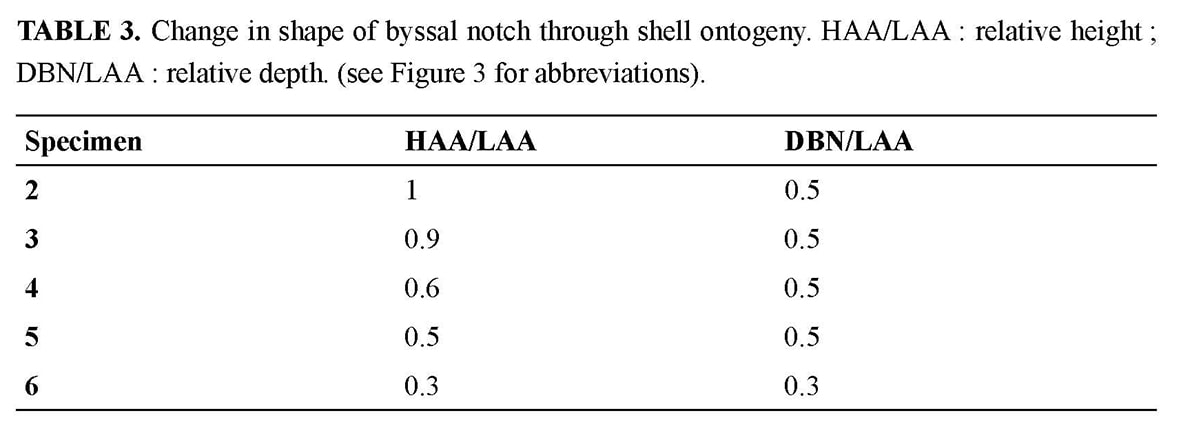
HOLOTYPE (Figures 4–7, Tables 2–3, specimen 6): Shell irregular, distorted by adaptation to the hard substrate (Figure 4a–b). In spite of distortion, right valve roughly flat up to a height of 11–12 mm (Figure 5a), and left valve convex. Closed valves without any gape other than the byssal notch. Auricles dissymmetrical with a byssal notch under the anterior auricle of the right valve.
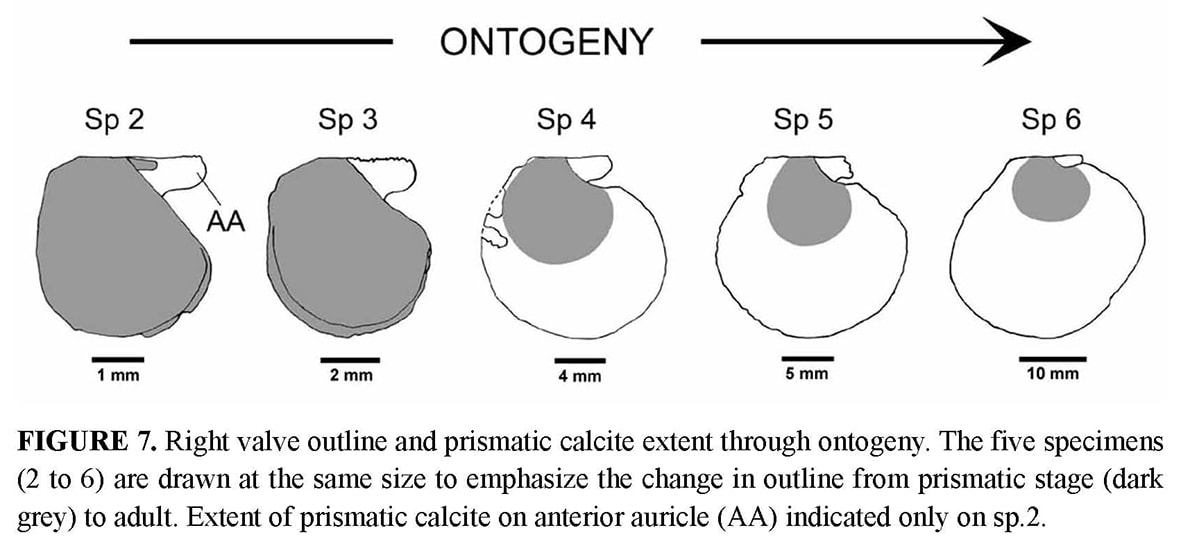
Anterior auricle (5.4 mm) slightly shorter than the posterior (5.9 mm). Posterior auricle poorly demarcated from disk, with only a shallow flexure between their surfaces, and no clear limit on the oblique posterior edge. Byssal notch without ctenolium hidden by the dorsal overlap of its ventral edge and thus observable only in dorsal view (Figure 6). Antero-dorsal edge of the disk (ventral edge of the byssal notch) sinuous, proximally leaving the depressed byssal fasciole uncovered, and distally extending slightly above the lower edge of the anterior auricle (Figure 5b). Dorsal expansion of this edge concomitant of a peculiar growth stage marked on the disk by a strong undulation just after a very sharp growth line at a UPD of 6 mm (Figure 4c).On both valves, disk and auricles with a dense commarginal ornamentation of ridges regularly spaced, and transforming to lamellae. On the left valve, ornamentation starts from the beak, covering disk and auricles. Interval between ridges or lamellae increasing with shell growth, from about 0.1 mm at a UPD of 12 mm, to 0.3 mm at least near the ventral edge. On the right valve, the outer prismatic layer terminates at a 10 mm UPD, and the same ornamentation as on the left valve then replaces the faint and narrow commarginal fluting. PARATYPES (Figures 2–3, 5–12, Tables 2–3, specimens 2 to 5): The type series constitutes an ontogenetic sequence (specimens numbered by increasing size), which offers the opportunity to follow some changes in shell morphology during growth.
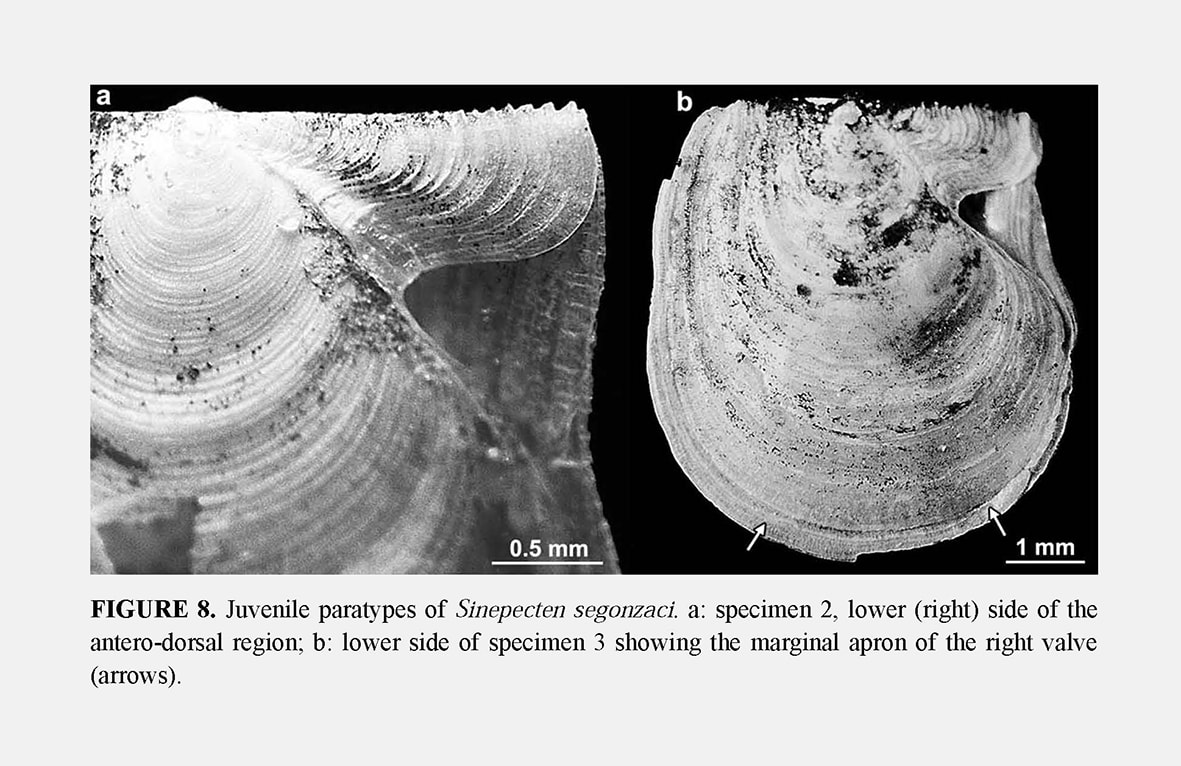
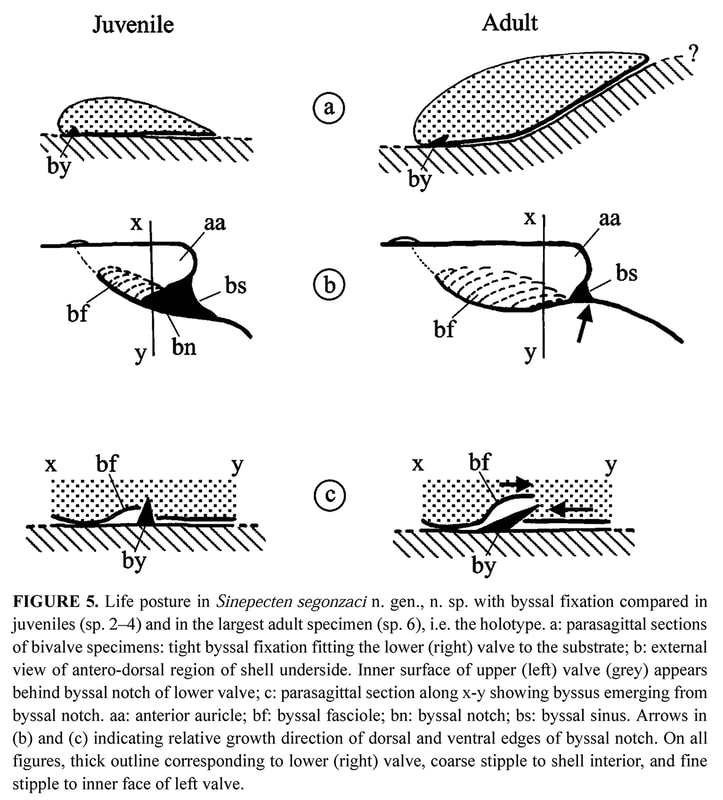
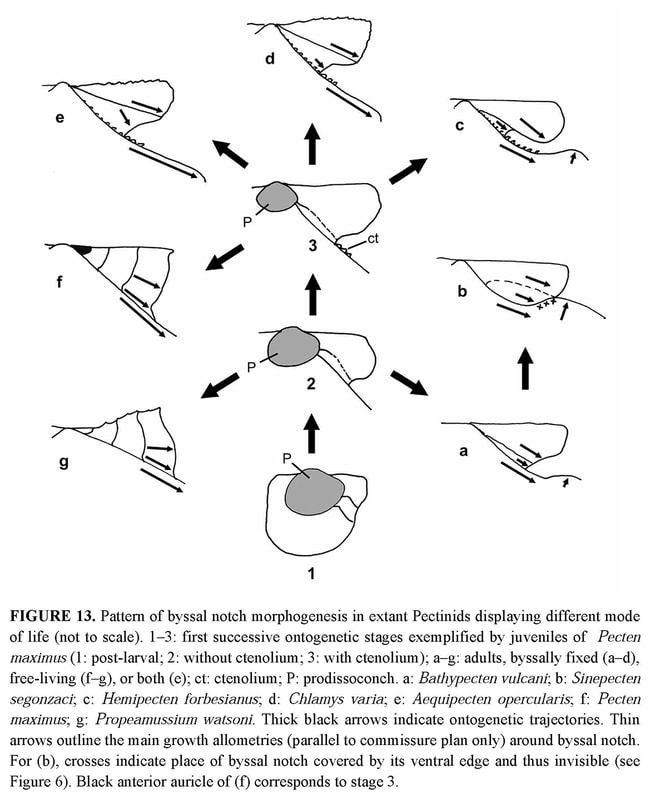
Shell outline: The outline of the shell in the commissure plane is the first character obviously changing through ontogeny (Figure 7), from higher than wide in the juvenile specimens 2 and 3 to as high as wide in specimens 4 and 5, and finally wider than high in the holotype. In the juvenile prismatic stage illustrated by specimens 2 and 3, when the shell is closed, the disk margin of the right valve is upturned and adpressed against the inner face of the left valve margin (Figure 2b–c). The line along which the margin of the right valve is flexed corresponds to a line of lower mechanical resistance, so that the marginal apron detaches as soon as the valves are separated. This character, which is common in Propeamussiinae (Schein 1989; Morton & Thurston 1989), disappears after the prismatic stage (specimens 4 to 6).
Auricles: None of the specimens has both its auricles intact, but their dissymmetry can be verified by reconstruction of the complete outline. The anterior auricle is slightly longer than the posterior in specimens 2 to 4, whereas it seems to be the opposite in the holotype (Table 2). In all specimens, the posterior auricle is poorly delimited. Its posterior edge is almost perpendicular to the dorsal edge in the juvenile specimens 2 and 3, and becomes increasingly oblique in specimens 4 to 6 (Figure 7).
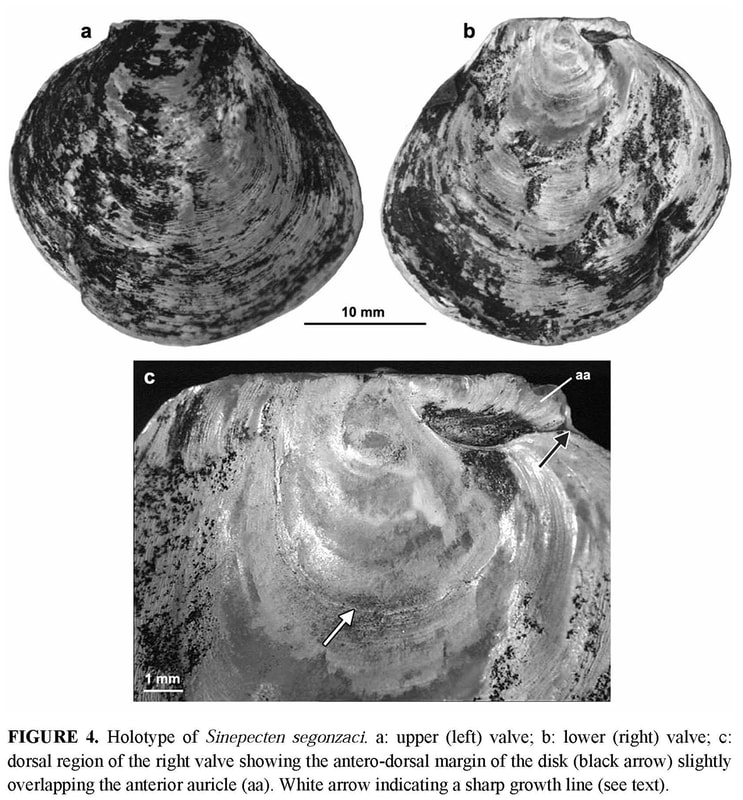
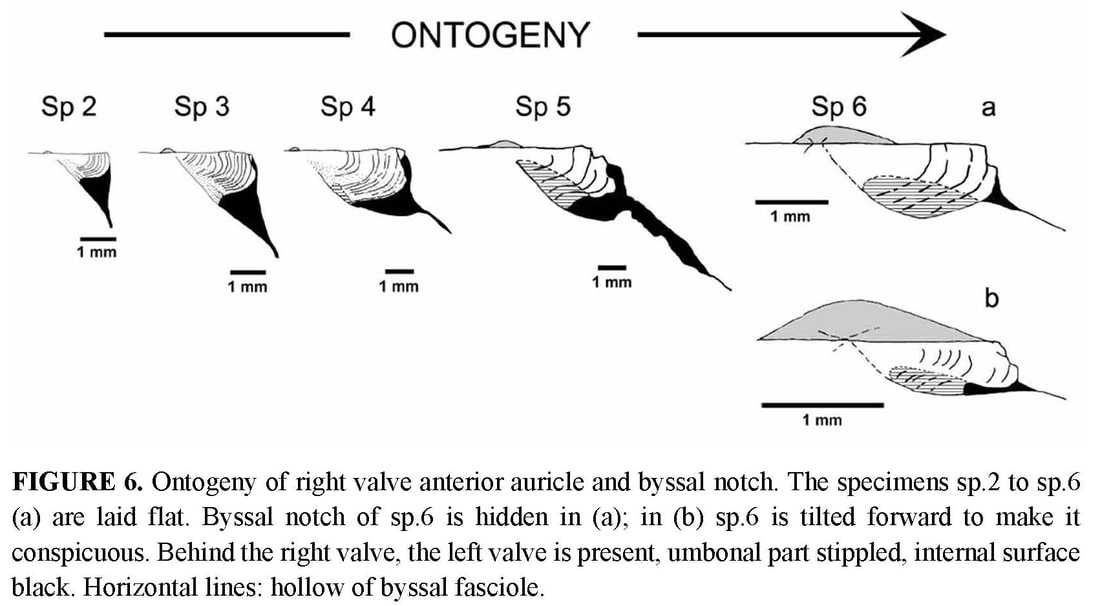
Byssal notch: On the anterior side of the right valve, the auricle and the byssal notch undergo a profound change (Figure 6). The byssal notch is triangular and widely open in the juvenile specimens (Figures 8, 9a), and begins to narrow after the end of the prismatic stage (Figure 9b). It is still quite visible on the specimen 5, but seems to have disappeared on the holotype (Figure 4c). Though the passage for the byssus is almost invisible in the holotype, it is not closed, but has undergone a change in size, shape and orientation. In the juvenile, it lies in the plane of the right valve, but later it sinks inwards with the ventral edge of the anterior auricle (i.e. the dorsal edge of the byssal notch), and in the holotype it has shifted to a plane more or less perpendicular to the disk plane so that the opening is dorsally directed (Figures 5b–c, 6). On the inner face of the specimen 4, there is a fold in the proximal end of the byssal notch (Figure 9b). This is probably induced by the ventral edge of the auricle beginning to move inward. In the holotype, this edge is covered by the antero-dorsal edge of the disk (Figure 4c). The byssal notch is not only narrowed by the overlap of its ventral edge, but also shortened by the byssal fasciole growing forward (Figures 5b, 13). The measurements show clearly the gradual decrease of height with growth, and the sudden drop of depth in the holotype (Table 3). This morphological change starts after the prismatic stage and is achieved in the largest specimen only.
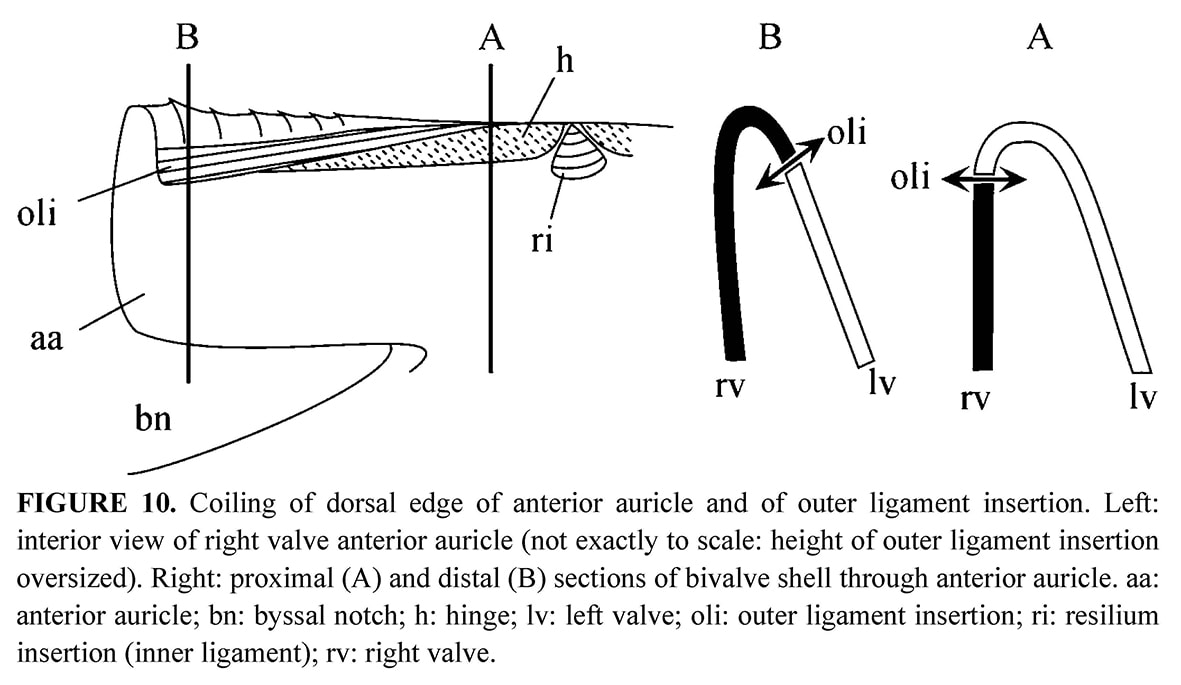
Ligament areas: On the internal face, the resilifer in which the inner ligament is attached, is prosocline (Figures 9b, 9e). The outer ligament, which prevents shearing or rotational movements of the valves, is attached along the dorsal edge of auricles. On each auricle, the dorsal edge is coiling from the beak to the distal extremity, and this is achieved as follows: the left valve beak overhangs the right valve, but it is opposite in the anterior end, where the dorsal edge of the right valve curves over the left valve (Figure 10). This coiling is absent in the juveniles, and owing to the poor preservation of the other specimens, it was not possible to ascertain which valve curves over the other in the posterior auricle. It is probably the right valve over the left, and to a less extent than anteriorly as in most pectinids examined in comparison, i.e., Pecten maximus Linné, 1758 and Chamys varia Linné, 1758 for example. On the opposite, in the deep-sea Propeamussium lucidum (Jeffreys, 1873) described by Morton & Thurston (1989) the left valve curves over the right at the distal end of the posterior auricle, and more markedly than at the end of the anterior auricle. In all cases, the same valve curves over the other at the anterior and posterior ends of the dorsal margin. This curvature is probably necessary to make the locking device efficient.
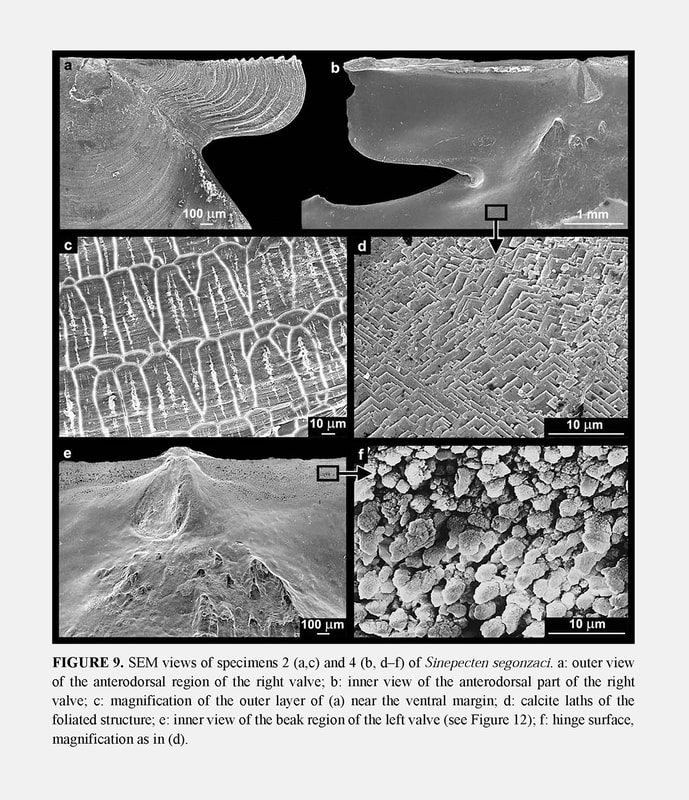
Hinge: Immediately ventral to the outer ligament, two flat areas are separated by the resilifer on each valve: each auricle has one such area which stretches along the dorsal margin and tapers almost to nothing at its distal extremity (Figures 9b, 9e, 10). The flat areas are edentulous, but their surface is minutely roughened, and therefore opposed to shearing. In fact, SEM observation of this toothless hinge shows that between the valves the calcite crystals clash by their points (Figure 9f). The rod shape of the crystals and their more or less upright orientation in the hinge contrasts with their tablet shape and their arrangement parallel to the depositional surface outside the hinge where the typical foliated structure covers the inner surface of the shell (Figure 9d).
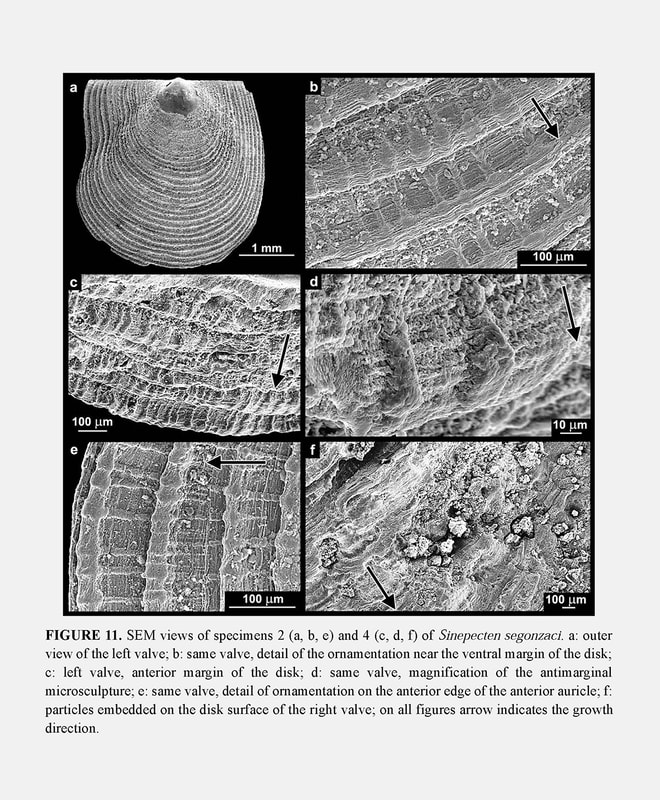
Ornamentation of left valve: The commarginal ornamentation affects only the external surface of the shell (Figure 11a). The interval between the ridges or lamellae covering the left valve is about 0.1 mm near the ventral margin in juvenile specimens 2 and 3 (Figures 11a–b,e), and reaches 0.2 mm in specimens 4 and 5 (Figure 11c). The spacing between ridges tends to increase with growth and exceeds 0.3 mm in the holotype. The increase is not regular, as measurements have shown significant variations, obviously related to frequent growth disturbances. The components of ornamentation are shaped as ridges or lamellae depending on their location. Ridges form near the beak and in the middle of the disk (Figure 11b), both places where growth rate is high. Lamellae form in the disk periphery (Figure 11e) and near the limits between disk and auricles, both places where growth rate decreases. Anyway, both ridges and lamellae represent a relative slowing of growth rate as evidenced in SEM views by the bunching up of growth lines on them. SEM observation also allows clarification of the details of the antimarginal microsculpture, which is superimposed on the ornamentation. In the juvenile, extremely fine and low antimarginal riblets are arranged regularly along the intervals between the commarginal ridges (Figure 11b–e). The riblets distally cross the ridges, on which they induce a regular pattern of close-set thickenings (Figure 11e). When the ridges turn to lamellae, these thickenings become sharper and higher distally (Figure 11d).
Ornamentation of right valve: Juvenile specimens 2 and 3 illustrate the prismaticstage. Their right valve is externally covered with a layer of calcite prisms extending to the disk margin. On specimens 4 and 5, the prismatic stage is a little shorter than on the holotype: 8 to 9 mm for specimen 4 and 9 mm for specimen 5, versus 10 mm for specimen 6. The array of prisms forms a pattern of recurrent, commarginal sequences (Figure 9c) corresponding to the concentric prismatic structure described by Schein (1989). As a consequence of this superficial microstructure, the right valve seems macroscopically to be unornamented in the prismatic stage. However, it displays weak and narrow commarginal flutings derived from the prismatic microstructure, and several irregular commarginal undulations (Figure 8a). As in the holotype, the specimens 4 and 5 develop on their right valve the same ornamentation as on the left one after the prismatic stage. The right valve ornamentation is thus late when compared to the left valve. The delay is longer on the disk than on the auricles, and in the latter, the anterior auricle is the more advanced. This is illustrated by the juvenile specimen 2 with an anterior auricle displaying relatively sharp commarginal ridges (Figure 8a). The ornamented part of the auricle lies outside the proximal prismatic area, which is very reduced (Figure 7).
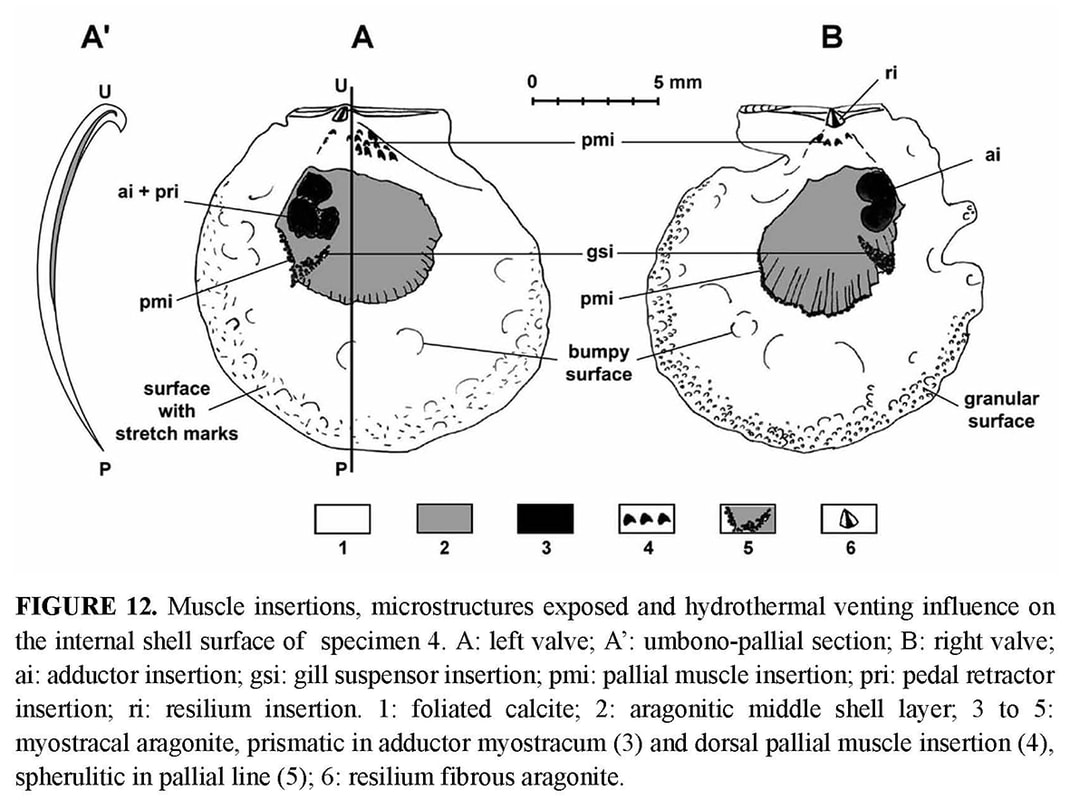
Aragonitic layer: On the inner face of both valves, there is an aragonitic layer outcropping, which is limited dorsally by the distal edge of the foliated calcite inner layer and ventrally by the pallial line (Figure 12). Its radiate organisation is obvious, with narrow bands radiating from the beak. SEM observation shows that the bands are alternatively light (more emissive) and dark (less emissive).
Muscle scars: The adductor muscle scar is different in the right and the left valves (Figure 12). On the right valve, the adductor scar is bean-shaped, with two adjacent parts placed one above the other. It is clearly defined, and the size of the two parts is equivalent. On the left valve, the scar is larger and less clear, but three sub-equivalent parts seem to be outlined. Two of them are posterior and superposed, and the third scar lies anterior to the ventral one. All three correspond to the muscular sectors responsible for the three basic functions in byssally attached pectinids: rapid adduction of valves (“quick” striated muscle), slow adduction (“catch” smooth muscle), and byssal attachment (byssus and foot retractor). Unfortunately, given the poor preservation of specimens, it was not possible to precisely identify where the corresponding muscles inserted.
Larval shell: The larval shell, which was either destroyed by corrosion, or coated with ferromanganese deposits could not be observed. Some remains on the left valve of specimen 2 allow us to estimate that the antero-posterior diameter is about 215 μm as in the larval shell of Bathypecten vulcani which has a non-planktotrophic type of development (Roux et al. 1989).
In life habit, S. segonzaci is byssally attached, and its valve lower flat valve is adpressed against the substrate (Figure 5). Hydrothermal environment: The five specimens of the type series of S. segonzaci were collected at the Barnacle vent site (PACMANUS area, Field E) on glassy basalt, among sessile barnacles, where the temperature ranged between 2.8°C at the periphery of hydrothermal fields and about 40°C in vent fluid. This site is a group of small chimneys from 6 to 8 m height, either inactive or emitting a black fluid (260 to 270°C). The vent fauna is established around these edifices, between cracks emitting a transparent fluid measured at 5 to 40°C (Hashimoto et al. 1999).
Kai Kai site is one of the two large diffuse vent sites of the Field D where chimneys are absent but where there are large diffuse vents with cracks emitting strong shimmering water at 30°C (Hashimoto et al. 1999). A single dead juvenile valve (specimen 1) was collected in a sediment area, at some tens of meters of the Kai-Kai vent site, and thus, outside of the vent influence. The outer shell surface of the specimens shows the fingerprint of the hydrothermal environment in which they grew, i.e., an aggressive and unstable environment, with fluids rich in metal ions ready to precipitate over the shell. The specimens all had their upper (left) valve coated with a ferromanganese black deposit, thicker in umbonal area. The black deposit also affected the lower (right) valve, where it was thinner and less regularly distributed (Figure 4). Damage to the shells increases with growth and size of the shell. When the individuals are small (specimens 2 and 3), the shell is blackened but its outer surface only is affected, and the outline is regular. In contrast, the valves of the larger specimens are affected on both sides, as the outer disturbances are reflected on the inner shell face. For example, specimens 4 and 5 are more or less deformed by a great number of bumps, and moreover the periphery of the disk is densely covered with countless particles which were trapped along the growing edge and became embedded into calcification (Figure 11f). On the left valve, the inner surface is densely scattered with marks resembling stretch marks; the inner surface of the right thinner valve is granular (Figure 12). The thinness of the growing edge explains why the inner face reflects the incorporation of foreign particles on the outer face. The high density of particles along the ventral margin of specimens 4 and 5 reflects their density in the ambient seawater. Finally, in the holotype, besides the black coatings, the shell is entirely distorted and seems even to have undergone a torsion, when compared to the younger specimens. This is obviously induced by its adaptation to the irregular substrate, but a reaction to the toxicity of fluids could also be involved. As a result of their small size and of the black ferromanganese deposits covering the shells (as well as external surfaces of the barnacles), these pectinids are not easily visible, since they are attached to a black basaltic substrate. Adaptive and phyletic affinities through comparative ontogeny
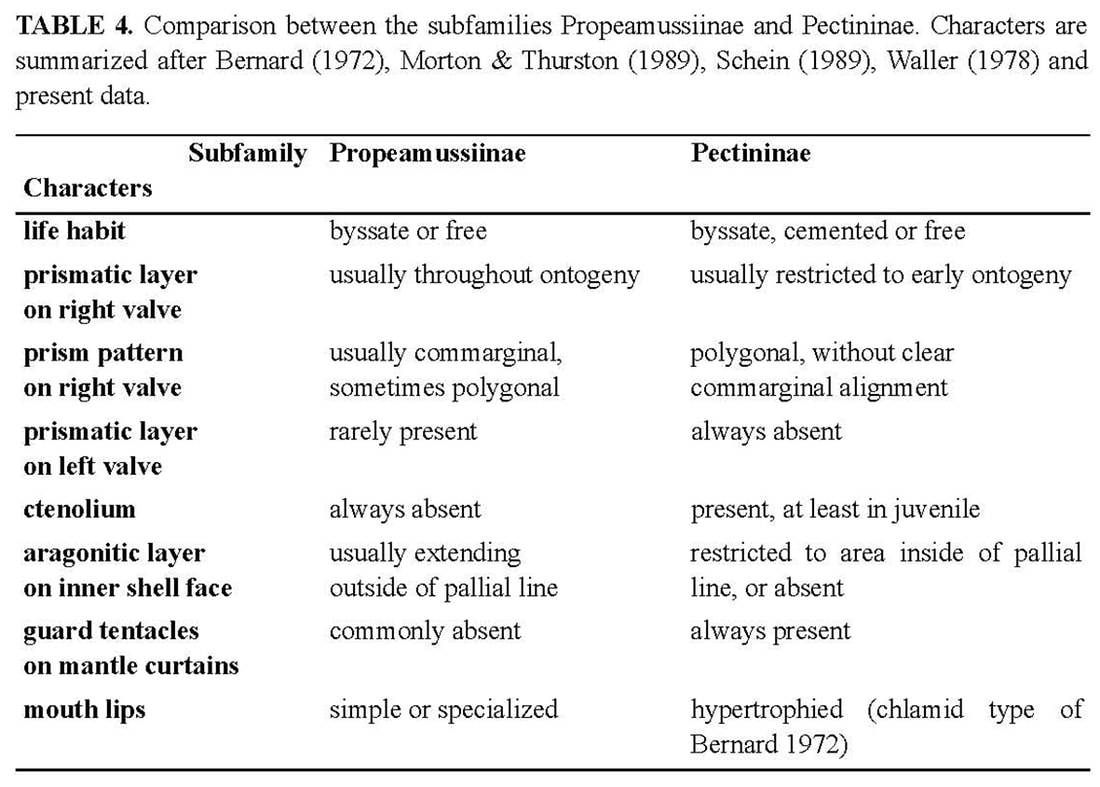
The classification of Sinepecten segonzaci in Propeamussiinae raises some uncertainties. As indicated above (see Introduction), the prevalent classification recognizes a superfamily Pectinoidea, divided into a number of families including the Pectinidae and Propeamussiidae (Waller 1978). However, Schein (1989) questioned a separation of these two families and proposed to downgrade them at subfamily rank. Obviously, the ctenolium is the main criterion for separating these subfamilies (Table 4). The other distinctive characters generate many intermediate patterns as in Sinepecten.
The shell characters common to Sinepecten and Propeamussiinae are (1) the absence of a ctenolium, (2) the commarginal arrangement of prisms on right valve, which corresponds to the concentric prismatic structure described by Schein (1989). This type of arrangement occurs in most members of Propeamussiinae, in which it persists throughout ontogeny. On the contrary, in Pectininae, the most common arrangement of prisms is polygonal without clear commarginal alignment, corresponding to the pavimentous prismatic structure described by Schein (1989). Two shell characters are common to Sinepecten and Pectininae, (1) a prismatic stage restricted to early ontogeny, (2) an aragonitic layer restricted to area inside the pallial line. It has to be emphasized that Sinepecten is the first known example of concentric prismatic microstructure restricted to early ontogeny. Sinepecten segonzaci is therefore an intermediate taxon, exactly opposite to Lissochlamis exotica (Dillwyn, 1817) from the littoral zone of west Africa, which is ctenoliate with prismatic calcite persisting on the right valve throughout ontogeny. The new taxon thus supports the concept of a single family, i.e., the Pectinidae, bringing together ctenoliate and non-ctenoliate genera (Schein-Fatton 1987; Schein 1989). The absence of a ctenolium is a plesiomorphic character (Waller 1978), and therefore not a proper base for a major systematic unit (i.e. Propeamussiidae), while the presence of a ctenolium is an innovation justifying the distinction of the subfamily Pectininae among the Pectinidae. Therefore, at present, the distinction between two subfamilies will be relevant only if the ctenolium is considered as a major derived character (synapomorphy) appearing in the Triassic and identifying a new lineage with peramorphic trends, i.e., the Pectininae. Peramorphy is the product of heterochronic processes driving to derived characters (see below, after the comparison of Sinepecten segonzaci with fossil Pectinoidea). In such an analysis, the extent of the outer prismatic calcite and of the inner aragonite layers would result from heterochronic processes working in both subfamilies, but particularly in the Propeamussiinae which belong to a more ancient lineage. Their greater age is suggested by their affinities with the paleozoic Pernopecten and Euchondriidae (Upper Devonian to Upper Permian), in which the origin of the superfamily Pectinoidea could likely lie (Waller 1978, 1998). However, soft parts should also be taken into account. Waller (1978), for example, used differences in lips and mantle tentacles to separate Pectinidae from Propeamussiidae. In fact, such anatomical data are still so scarce in the latter (Knudsen 1970; Bernard 1972, 1978; Morton & Thurston 1989) that their relevance at the family level is questionable. For example, the most detailed study at present (Morton & Thurston 1989), which deals with the functional morphology of Propeamussium lucidum (Jeffreys, 1879), demonstrates that many soft part characters prove to be highly adaptive. In this carnivorous deep-sea species, filter-feeding is impossible and the ctenidia are of a new type, wholly subserving a respiratory function. Labial palps are vestigial, but large mouth and lips form a capacious buccal sac, into which preys are probably pushed with the foot. It is thus justified to question the reliability of such characters for systematic and phylogenetic purposes. Anyway, it was not possible to observe the distinctive characters of soft parts through the black-coated shells of Manus specimens. The byssal notch of Sinepecten segonzaci is unique among Propeamussiinae. In the holotype of B. vulcani, the ventral edge of the byssal notch is sinuous (Schein-Fatton 1985), as in the specimen 4 of S. segonzaci, which is about the same size (14 mm height) and in the early stage of the dorsal extension process. When S. segonzaci reaches 27 mm, i.e., almost twice the height of B. vulcani, the byssal notch is finally reduced to a narrow and short slit covered by its ventral edge. From a morphofunctional view, the byssal notch is the outer portion of a functional device in which the foot and its musculature are the main actors. The latter are reduced in pectinids, as in all monomyarian bivalves. The reduction is greatest in free-living species, with only the adductor impression on the inner face of each valve. This large muscle is divided into two components: a smaller smooth (catch) portion responsible for a prolonged adduction of the valves, and a larger striated (quick) portion responsible for a rapid adduction of the valves, as during swimming and cleansing actively (Chantler 1991). But in byssally attached species, the foot is concerned with byssal secretion, and retains the left posterior pedal retractor needed for pulling the animal down on the byssal attachment (Yonge 1981). The impression of the pedal retractor muscle is present on the left valve, close to the adductor. It appears thus that muscle impressions could give some clues for life habits, as exemplified by Hemipecten forbesianus Adams & Reeve, 1849. Hemipecten forbesianus lives attached to coral colonies in the south-western Pacific (Yonge 1981; Dijkstra & Marshall 1997). With a ctenolium and a very deep byssal notch, it greatly differs from S. segonzaci. However, both species have some common characters, resulting probably from similar constraints in byssal attachment: a compressed shell without radial ornamentation, a flat and thin right valve, a curved byssal notch, and particular muscular impressions on the left valve. Yonge (1981) described the byssal attachment of H. forbesianus to the smooth under surface of a coral colony, the shell conforming perfectly to the coral. The byssus of H. forbesianus is hypertrophied and extends through the byssal notch in an obliquely upward (dorsal) direction as in S. segonzaci (Figure 5), and moreover, it has also been pushed far forward. On the left valve, muscle proportions are unusual. Yonge (1981) emphasizes the immense hypertrophy of the left retractor, larger than the adductor in which the two portions are separate with the smooth part only a little smaller. In S. segonzaci, there are three subequal muscular impressions in the left valve (Figure 12). This suggests that the pedal retractor is much smaller than in H. forbesianus, and that the smooth and striate portions of the adductor are subequal as in the latter, indicating that “prolonged adduction must be as important as cleaning contractions” (Yonge 1981: p. 30). Yonge (ibid.) considered also that in H. forbesianus “probably the valves do not gape widely when the adductor and pedal retractor relax”. This could be the same in S. segonzaci, and would be an advantage in an environment loaded with mineral particles.
Concerning life habit, S. segonzaci seems to live byssally attached throughout its life, like H. forbesianus and Pedum spondyloideum (Gmelin, 1791) (Yonge 1967, 1981; Waller 1972). All three of these species display a very peculiar byssal notch and a hypertrophied left pedal retractor. But P. spondyloideum differs from the other two in growing on a living substrate of coral in which it ultimately becomes encased. The constraint on the development of H. forbesianus and S. segonzaci is different. Both species exhibit a byssal notch shaped by the converging growth of its edges (Figure 13b–c) through which the byssus extends in an obliquely upward (dorsal) direction. The convergence starts very early in H. forbesianus with the ventral growth of the auricle, but there is a sharp growth gradient between the byssal fasciole and the dorsal and ventral edges of the byssal notch. The result is a deep, curved slit, with subparallel edges, the ventral one being ctenoliate. In S. segonzaci, the convergence starts later at the end of the prismatic stage, with the forward growth of the byssal fasciole and the upward growth of the ventral edge. However the process runs farther than in H. forbesianus, driving the non-ctenoliate ventral edge to overlap the other, so that the short passage for the byssus is dorsally directed. Thus, starting from the juvenile stages (Figure 13, stage 2 or 3), the interplay of growth rates in the different parts surrounding the byssal notch will generate a more or less narrowly adapted morphology, according to the ontogenetic pathways in the Pectinidae. If the allometries of the early stage are not appreciably modified through ontogeny, there will be no sharp specialization (Figure 13d–e) as in Chlamys or Aequipecten, in which the adult can generally change its habit. If the differences between growth rates in dorsal and ventral parts of the auricle are cancelled (Figure 13f–g), two opposite specialisations become possible: attachment becomes permanent as in cemented forms (i.e., Hinnites), or disappears as in free-living Amusium, Propeamussium or Pecten. If they are modified and/or if new allometries are added, it is the byssal attachment that specializes, as in Hemipecten and Sinepecten (Figure 13b–c). Among the three genera studied here, Bathypecten and Catillopecten follow the first process (Figure 13a) throughout life; they keep a medium-sized byssal notch without clear functional specialization, as Chlamys and Aequipecten. During the juvenile stage, Sinepecten is the same way but later goes on to the third process (Figure 13b), when auricle and byssal notch undergo a profound change and become specialized in byssal attachment. It is only in the fossil record and among paleozoic or mesozoic extinct families that a byssal notch more or less similar to that of S. segonzaci can be found. The taxa retained belong to the superfamily Pectinacea (= Pectinoidea at present) taken into account by Hertlein et al. (1969). In chronological order, several examples can be quoted: — in the Pterinopectinidae, Pterinopecten (Upper Silurian to Upper Devonian) is very similar to B. vulcani, as underlined in the original description of the latter (Schein-Fatton 1985, 1988) with a byssal notch shaped as a curved narrow slit; — in the Aviculopectinidae, Claraia (Lower Triassic) with a “subauricular notch narrow, widening inward”(Newell 1969: p. N337); — in the Oxytomidae, Maccoyella (?Late Jurassic to Lower Cretaceous) with “subauricular notch narrow, oblique, clearly seen only from interior of valve, or possibly absent in some specimens” (Cox 1969a: p. N346); — in the Buchiidae (Upper Triassic to Cretaceous), the “right anterior auricle (is) well demarcated, with deep acute subauricular sinus in most genera and extending out of plane of valve margins toward left valve in some forms” (Cox 1969b: p. N374) as in Hokonuia (Upper Triassic) and Malayomaorica (Upper Jurassic). Such an enumeration shows that the type of byssal notch present in S. segonzaci has been achieved on several occasions during the evolution of Pectinoidea. Its occurrence in different taxa and in different periods indicates the colonization of ecological niches requiring the same attachment device. Heterochronic processes can maintain juvenile characters in the adult (paedomorphy) or, on the contrary, induce derived characters in the adults (peramorphy) (Gould 1977; McKinney 1988; McNamara 1995). Thus, when paleontological and/or molecular data are missing as in Propeamussiinae, the two hypotheses which can be considered for a taxon cumulating juvenile characters in the adult, are either ancestry (antiquity and phylogenetic relationship) or paedomorphy.
At the time of its discovery, B. vulcani was interpreted as a particularly archaic taxon (living fossil) in reference to paleozoic fossils (Schein-Fatton 1985, 1988). It was then a widely held view, and the numerous endemic, new invertebrate species discovered around the hydrothermal vents were considered as Paleozoic or Mesozoic relicts (Newman 1985). However, molecular phylogenies tend to invalidate this hypothesis (Mc Arthur & Tunicliffe 1998, Little & Vrijenhoek 2003, Geiger & Thacker 2005), at least regarding the major taxa of the chemosynthetic communities of hydrothermal environment. Sinepecten segonzaci is probably an occasional and peripheral element of these communities, like B. vulcani, in which no evidence of trophic reliance on endosymbiotic bacteria could be found (Le Pennec et al. 2003). For most of the Propeamussiinae, a paedomorphic trend related to the scarcity of trophic resources in the abyssal environment has also been suggested (Schein-Fatton 1987). Could B. vulcani illustrate an extreme degree of this paedomorphic trend, despite the abundant trophic supply in the hydrothermal environment? Indeed, paedomorphy can be defended against archaism (ancestry) hypothesis only as an opportunistic response to environmental instability (Gould 1977). The limited duration of the prismatic stage in S. segonzaci indicates a degree of peramorphy which the other Propeamussiinae do not reach. In regard to Bathypecten and Catillopecten, the same peramorphy affects both ornamentation and byssal notch. The two species colonizing the hydrothermal environment thus take place at the opposite ends of the heterochronic gradient along which the three genera range. The derived characters of S. segonzaci tend to invalidate the assumption of a paedomorphic trend related to instability in hydrothermal vent environment, thus supporting the ancestry hypothesis for B. vulcani. Remarks on biogeography
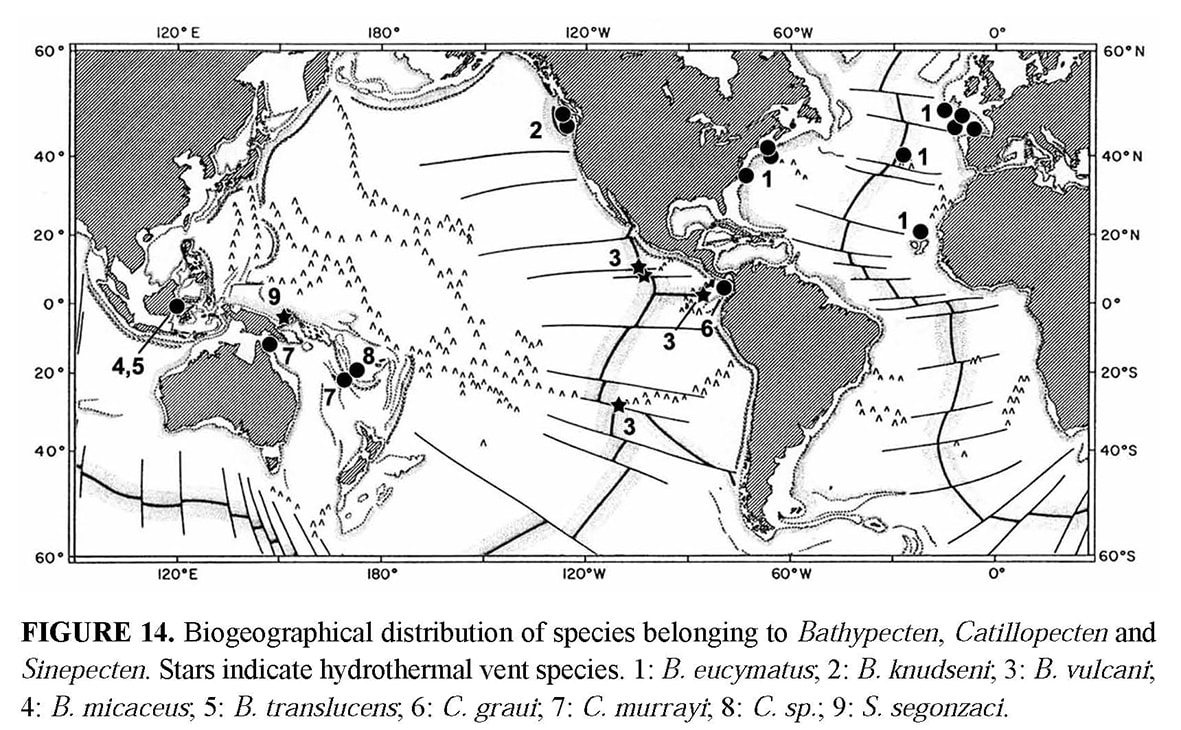
Among the three genera Bathypecten, Catillopecten and Sinepecten, Bathypecten is the most cosmopolitan. Bathypecten vulcani has been collected on hydrothermal vent environments of the East Pacific Rise at 9° 50’N, 13° N and Galapagos at depths ranging 2470–2650 m (Schein-Fatton 1985, 1988; Roux et al. 1989; Le Pennec et al. 1988, 2002; Mullineaux et al. 1998). Many specimens in good preservation have been recently retrieved on the Pacific Antarctic Ridge from hydrothermal environment (PAR5 cruise, March 2005, 31° 51.88’ S – 112° 02.69’ W, 2335 m, Schein unpublished data). Bathypecten eucymatus (Dall, 1898) is an amphiatlantic abyssal species collected at depths ranging 1950 to 4839 m and its size hardly exceeds 10 mm. The distribution drawn up by Schein (1989) has to be completed in the north-eastern Atlantic with new findings from Galicia Bank at depths ranging 985–1125 m (Dijkstra & Gofas 2004) and from North of Cape Verde Rise at depths ranging 3107–3188 m (French cruises EUMELI, Ifremer, unpublished data).
According to Schein (1989) several species classified in the genus Cyclopecten and displaying a poorly demarcated posterior auricle and a smooth or undulated surface (Dautzenberg & Bavay 1912; Bernard 1978), belong evidently to Bathypecten: B. knudseni (Bernard) has been collected in the Eastern Pacific off Oregon and Vancouver Island from 44°N to 50°20’N and at depths ranging 220 to 2900 m; B. micaceus (Dautzenberg & Bavay) and B. translucens (Dautzenberg & Bavay) come from Makassar Strait at a depth of 1301 m. Catillopecten is a monospecific genus established by Iredale (1939) for C. murrayi (Smith), a species for which the single known specimen has been collected by the “Challenger” in north-eastern Australia, East of Cape York, at a depth of 2520 m (Smith 1885), and has a UPD of 14.5 mm and a APD of 15.5 mm. Two new specimens were collected off New Caledonia (CALSUB cruise, February 1989, dive 17, 21° 25’ S – 168° 24’ E) allowing to observe the non-planktotrophic larval shell. One specimen is a single left valve from a depth of 1700 m, and the other is a bivalve specimen that was attached to a dead shell of Nautilus from a depth of 1865 m. Two specimens undoubtedly belonging to the genus Catillopecten have recently been retrieved from the North Fiji back-arc Basin (TUIM6MV Cruise, May 2005) in areas without thermal vent activity: a single broken left valve from a depth of 2706 m (18° 49.04’ S – 173° 29.37’ W), and a bivalve juvenile from a depth of 1991 m (16° 59.48’ S – 173° 54.89’ W). The distance separating these localities from the Manus Basin where S. segonzaci was collected is thus relatively short. Described by Knudsen (1970) from the Gulf of Panama, eastern Pacific, at depths ranging 3188 to 3270 m, Cyclopecten graui Knudsen displays both commarginal undulations and narrow folds, and is to be transferred in the genus Catillopecten according to Schein (1989). Cyclopecten squamiformis Bernard from the north-eastern Pacific at depths ranging 2030 to 3188 m (Bernard 1978), is similar to Catillopecten in having a poorly demarcated posterior auricle and commarginal lirae (= fine ribs) on both valves. However, as there are also radial lirae on the left valve, it seems better to leave it in the genus Cyclopecten. Catillopecten murrayi, C. graui, B. translucens and B. micaceus have each been described from a single specimen, and moreover, the specimens were not complete in the two Bathypecten species. Additional specimens are necessary to complete their description and refine the comparisons. Bathypecten translucens and B. micaceus which were retrieved from the same station and are relatively similar, could belong to the same species. Thus, in the present state of knowledge, the three genera comprise eight species distributed in the central Atlantic, Bay of Biscay, tropical and north-eastern Pacific (Figure 14). The distribution of Bathypecten species on both sides of the central Atlantic and Pacific suggests that it could be inherited from the plate tectonic history, and more precisely from the Jurassic mesogean Tethys. Catillopecten and Sinepecten seem to be confined to the Pacific Ocean. The genus Bathypecten is principally from depths ranging 1000–3500 m although B. knudseni was collected from relatively shallow depths (220 m) and B. eucymatus from almost 5000 m. Catillopecten and Sinepecten seem to be restricted to intermediate depths ranging 1300 to 3300 m, outside abyssal plains and epibathyal environments. Therefore, the hypothesis of Bathypecten being the most ancient genus is also supported by the extent of its geographic and bathymetric distribution, which is the widest of the three genera. As in all the back-arc basins of the western Pacific, the hydrothermal vent site of the Manus basin is geographically isolated from vents along mid-ocean ridges (Van Dover et al. 2002; Desbruyères et al. 2005). Therefore, its colonization may have contributions from other sources, although Hessler & Lonsdale (1991) proposed that exchanges between the East Pacific Rise and western Pacific vent fauna might have occurred via the Kula Ridge during the early Cenozoic. Sinepecten segonzaci, like B. vulcani, lives on a vent site that is relatively close to the bathyal station where another species belonging to one of the two other genera is present. These observations suggest colonization of hydrothermal sites by local bathyal species. At present, more information on larval dispersal, and more sampling on the rocky bottoms where these fragile bivalves attach, are needed to validate the formulated assumptions.»
ELISABETH SCHEIN, 2006
|
|
«Type material
Holotype NSMT Mo-73697 and paratypes (4, NSMT Mo-73698-701), 03°43.62’S, 151°40.32’E, hydrothermal vent field, Manus back-arc basin, Papua-New Guinea, alive attached to rocks, 1674 m, 1998, submersible Shinkai 2000. Material examined
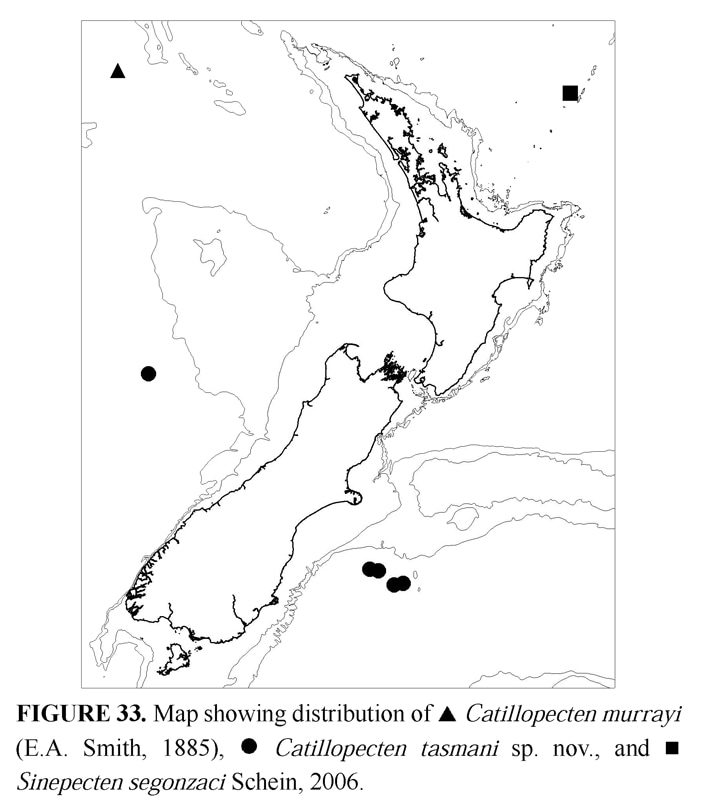
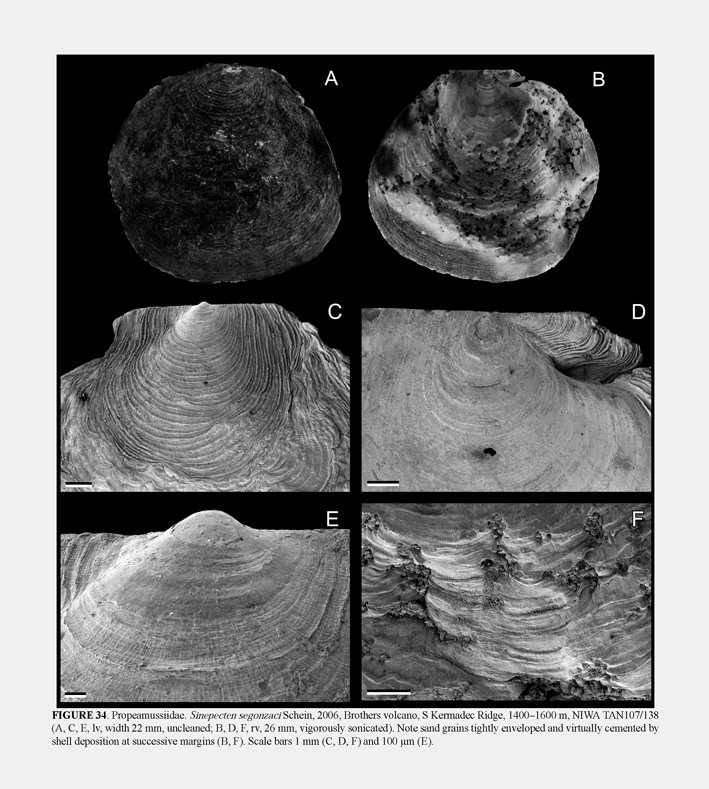
34º51.14'S, 179º02.74'E, Brothers volcano, S Kermadec Ridge, 1400–1600 m, 22 May 2001, RV Tangaroa, attached to volcanic rocks (1 pr, M.183888; 1 pr, ZMA Moll. 146829; 2 pr, NIWA TAN107/138). Distribution
Manus back-arc basin, Papua-New Guinea, and Brothers volcano, southern Kermadec Ridge (Fig. 33). Remarks
The present material is morphologically identical to the type species, although the specimens from the Kermadec Ridge have coarser and more widely-spaced commarginal ridges when the shell is larger than 10 mm in length (ca. 2 versus 6 per mm). The present specimens (height 21–24 mm: no ctenolium) were rather firmly attached to the substratum both by a weak byssus and such extremely fine and precise interdigitation of the growing margin of the right valve with the substratum that projections of the substratum are tightly enveloped and small particles are embedded in the shell (Fig. 34F). Grip on the substratum was so strong that specimens had to be carefully levered off with a knife to avoid breakage, but sufficiently weak that the specimens could be separated from it intact. The relatively weak byssus is evidently functional only in early ontogeny (height <9 mm), since grip by the dissoconch is substantially firmer. Schein (2006) suggested that irregularities in the shell were due to “damage” and suggested that it could also be a reaction to toxicity of fluids, but admitted that it was “obviously induced by its adaptation to the irregular substrate.” Schein further suggested that the high density of particles along the ventral margin reflects their density in the ambient seawater, but by contrast, we perceive their incorporation as due to the method of attachment to the substratum rather than incidental entrainment. This form of non-byssal attachment to the substratum is unique among Pectinoidea, perhaps transitional to full cementation.» DIJKSTRA, H. H. & B. A. MARSHALL. 2008. The recent Pectinoidea of the New Zealand region (Mollusca: Bivalvia: Propeamusiidae,Pectinidae and Spondylidae). Molluscan Research, 28 (1): 1-88, figs. 1-70. [p. 41, 42]
|
Sinepecten segonzaci Schein, 2006; H. H. Dijkstra & B. A. Marshall, 2008, The recent Pectinoidea of the New Zealand region, figure 34.
|